Mitochondrial Energy Metabolism & Vessel Wall Heterogeneity in Diabetic Atherosclerosis
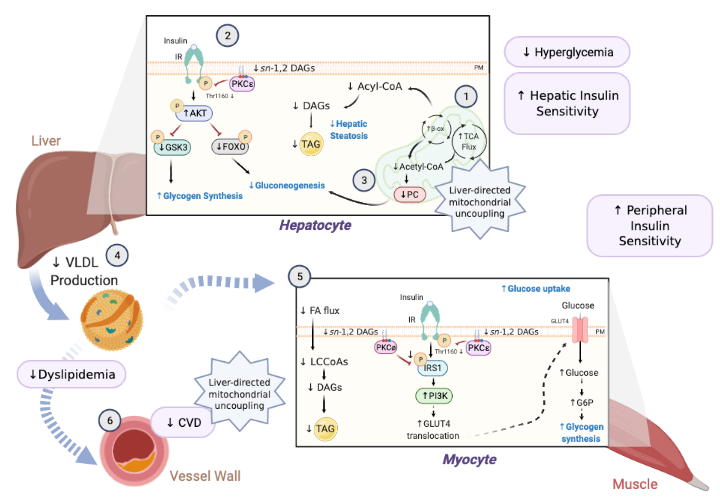
Figure 1: Therapeutic efficacy of liver-directed mitochondrial uncoupling agents (CRMP) for the treatment of cardiometabolic disorders.
Created with Biorender.com. Modified from PMID: 33545391, Figure 3.
Considerable evidence suggests that insulin resistance (and the resultant hyperinsulinemia) can explain the residual cardiovascular disease risk in diabetic patients; however, the underlying mechanisms remain incompletely understood. In particular, the field has been limited by a lack of appropriate animal models and technical methods needed to understand how the diabetic milieu (eg. hyperglycemia, hyperinsulinemia) may drive atherogenesis independently of changes in plasma cholesterol. We have previously shown that liver-directed mitochondrial uncoupling agents (controlled release mitochondrial protonophore, CRMP) can safely and effectively reverse dyslipidemia, fatty liver disease, and insulin resistance in rodent and nonhuman primate models of metabolic syndrome by increasing hepatic fat oxidation through a subtle sustained increase in hepatic mitochondrial inefficiency (Sci Trans Med, 2019; Aging Cell, 2021). Relevant to the projects outlined herein, we have recently demonstrated that CRMP reduces atherogenesis in a mouse model of atherosclerosis and metabolic syndrome (Fig. 1–2), at least in part, through systemic modulation of insulin sensitivity and reductions in hypertriglyceridemia (ADA 79th Scientific Sessions, 2020). Current projects in the lab aim to 1) further define the mechanisms by which mitochondrial uncoupling reduces the progression of atherosclerosis in mice and 2) leverage the insulin-sensitizing effects of CRMP to directly assess the role of hyperinsulinemia and insulin-resistance in driving diabetic atherosclerosis.
Fig. 2: En face oil red O staining of atherosclerotic plaques in Ldlr-/- mice treated with CRMP or vehicle control for 12 weeks.
In addition, we are developing and utilizing novel state-of-the-art metabolic tracer methods to characterize the regulation of macrophage immunometabolism during diabetic atherosclerosis, as the relationship between the inflammatory status and bioenergetic profile of plaque macrophages in vivo, as well as the impact on atherosclerotic development and stability, remains largely unknown. Collectively this work will provide meaningful insight into the mechanisms regulating diabetic atherosclerosis and will be critical for understanding the therapeutic utility of liver-directed mitochondrial uncoupling agents for the treatment of cardiometabolic disease. We also strive to utilize the comprehensive metabolic toolkit that we are establishing from these research projects (in combination with scRNA-seq and scATAC-seq) to interrogate a broader set of intimal cells (endothelial cells and vascular smooth muscle cells) during disease progression, allowing us to understand the functional relevance of vessel wall heterogeneity during cardiometabolic disease.
Relevant Publications
Relevant Funding
NHLBI (NIH) Pathway to Independence Award K99/R00 | Cellular and Molecular Mechanisms of Diabetic Atherosclerosis